How direct engagement with users shaped KeyGrip’s path forward
For Megan Moynahan, a biomedical engineer and founder of KeyGrip, the goal was clear—develop an implantable device to restore hand function for people with cervical spinal cord injuries (SCI). The science was strong, and the need was well-documented. But one crucial question remained: How would potential users and surgeons actually perceive it?
Through the Praxis 2024 SCI Incubate Program, KeyGrip gained direct access to Persons with Lived Experience (PLEX) of SCI and regulatory experts. These conversations reshaped their messaging, reintroduced a product feature they had ruled out, and provided a clear roadmap for entering the Canadian market.
Hearing directly from users changed everything
KeyGrip’s initial assumption was that anyone with SCI seeking more independence would want to improve their hand function. But through focus groups with PLEX members, they uncovered a more complex reality—potential users had concerns about surgical risks, recovery time, and the fear of losing existing function.
Rather than just highlighting the benefits, KeyGrip needed to address fears head-on, ensuring their messaging reassured users and provided clear data on the long-term impact.
“We always knew people valued hand function—but hearing their concerns in their own words made us rethink how we communicate our solution.”
Bringing a feature back into the design
One of the most unexpected takeaways was how users viewed different control methods. Initially, KeyGrip had dismissed voice activation, assuming it wasn’t a priority. But user feedback challenged that assumption.
This insight put voice control back on the table, influencing upcoming development proposals.
“People may not want to talk to their hands in public, but I was really surprised that they are open to voice activation at home, making it a viable control method once again.”
A Clearer Regulatory Pathway for Canada
Beyond product and messaging refinements, Praxis helped KeyGrip navigate the Canadian regulatory landscape, an area they hadn’t initially prioritized. Through mentorship and expert connections, they developed a clear plan for clinical trials and approvals.
This new perspective expanded KeyGrip’s market strategy, ensuring they could plan for approvals in multiple regions early on.
Insights that will shape KeyGrip’s next steps
Unlike traditional incubators with a set curriculum, Praxis adapted to what FMRK needed—whether that was regulatory guidance, market validation, or direct feedback from Persons with SCI Lived Experience (PLEX).
The biggest shift for KeyGrip wasn’t just in strategy—it was in how they approached decision-making. Conversations with PLEX members provided real-world perspectives that no market research could replicate, while regulatory guidance gave Megan and her team the clarity they needed to move forward with confidence.
“Every time I have a stakeholder conversation; it improves my thinking in a positive way. It helps me make decisions faster, with more confidence.”
One of the most profound realizations was that building an innovative product isn’t just about function—it’s about trust. People with SCI valued their independence and were naturally cautious about interventions. The challenge wasn’t just demonstrating that KeyGrip could improve hand function—it was proving that it wouldn’t compromise
what users had already adapted to.
Rather than simply accelerating their development, the greatest impact of Praxis was helping KeyGrip make the right decisions at the right time. Whether through PLEX insights, regulatory guidance, or 1:1 expert mentorships, every step brought them closer to a product that truly meets the needs of both users and clinicians.
“We need to collect the right evidence, not just to prove our technology works, but to help people feel confident in making the decision.”
About KeyGrip
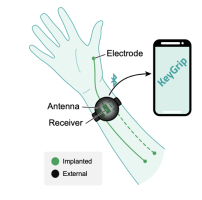
KeyGrip is a simple implanted stimulation system that provides lateral pinch to people with cervical spinal cord injury, enabling them to pick up and use small objects and giving them independence in eating, writing, and other activities of daily life. KeyGrip has 2-3 implantable electrodes that can be placed in an outpatient procedure to activate paralyzed muscles. Activation of the implant is achieved through externally placed components.


 How FMRK Uncovered a Critical Gap in UTI Testing and Pivoted Fast
How FMRK Uncovered a Critical Gap in UTI Testing and Pivoted Fast








 DeCell Technologies is a Canadian regenerative medicine company developing innovative biomaterials to treat chronic and complex wounds. Their flagship product, DermGEN™, is a human-derived decellularized matrix engineered to accelerate healing in wounds that fail to respond to standard care, including diabetic foot ulcers, burns, and pressure injuries. Unlike traditional grafts, DermGEN™ is gently processed to preserve the tissue’s natural regenerative properties without high-energy sterilization—resulting in a biologically active matrix that supports faster tissue repair and reduces complications. Already in use across multiple Canadian healthcare settings, DermGEN™ is transforming outcomes for clinicians and patients by offering an easy-to-use, effective, and accessible advanced wound care solution.
DeCell Technologies is a Canadian regenerative medicine company developing innovative biomaterials to treat chronic and complex wounds. Their flagship product, DermGEN™, is a human-derived decellularized matrix engineered to accelerate healing in wounds that fail to respond to standard care, including diabetic foot ulcers, burns, and pressure injuries. Unlike traditional grafts, DermGEN™ is gently processed to preserve the tissue’s natural regenerative properties without high-energy sterilization—resulting in a biologically active matrix that supports faster tissue repair and reduces complications. Already in use across multiple Canadian healthcare settings, DermGEN™ is transforming outcomes for clinicians and patients by offering an easy-to-use, effective, and accessible advanced wound care solution.